About one billion people worldwide are at risk for schistosomiasis — a debilitating disease caused by parasitic worms that live in fresh water and in intermediate snail hosts. A new study finds that the transmission risk for schistosomiasis peaks when water warms to 21.7 degrees centigrade, and that the most effective interventions should include snail removal measures implemented when the temperature is below that risk threshold.
The Proceedings of the National Academy of Sciences published the results, led by Emory University, the University of South Florida and the University of Florida.
“We’ve shown how and why temperature matters when it comes to schistosomiasis transmission risk,” says Karena Nguyen, a post-doctoral fellow in Emory University’s Department of Biology and a first author of the study. “If we really want to maximize human health outcomes, we need to consider disease transmission in the context of regional temperatures and other environmental factors when developing intervention strategies.”
The findings indicate that climate change will increase schistosomiasis risk in regions where surface water moves closer to 21.7 degrees centigrade, or 71 degrees Fahrenheit. The researchers also found, however, that implementing snail control measures decreases transmission but raises the temperature for peak transmission risk to 23 degrees centigrade, or 73 degrees Fahrenheit.
Co-first author of the paper is Philipp Boersch-Supan, an expert in ecological systems at the University of Florida and the British Trust for Ornithology.
Nguyen is a member of the lab of David Civitello, Emory assistant professor of biology and a co-author of the PNAS paper. The Civitello lab studies the ecological dynamics of disease, aquatics and agricultural ecology through a combination of experiments, field surveys and models.
“The control of schistosomiasis currently relies on treating infected people,” Civitello says. “However, there is renewed awareness that the ecological factors surrounding the disease also need to be considered. Our paper is a beautiful example of the potential power of uniting ecology with human disease interventions and control measures.”
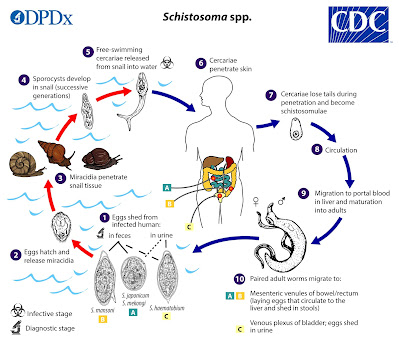
Click on graphic of the life cycle of the schistosomiasis parasite, above, to enlarge.
Schistosomiasis is one of the most devasting water-based diseases in developing countries, with more than 200 million people infected worldwide, leading to around 200,000 deaths annually. It is caused by Schistosoma parasites that have a complex life cycle. Freshwater becomes contaminated by the parasite’s eggs when infected people urinate or defecate in the water. After the eggs hatch, the parasites enter freshwater snails where they develop and multiply. More mature parasites are able to leave the snails and re-enter the water. These free-swimming parasites can then burrow into the skin of people who are wading, swimming, bathing, washing or doing agricultural work in contaminated water.
Children who are repeatedly infected can develop anemia, malnutrition and learning difficulties. Over the long term, the parasites can also damage the liver, intestine, lungs and bladder.
“Schistosomiasis is treatable — people can take a drug to get rid of the adult parasites in their bodies,” Nguyen says. “But in areas where schistosomiasis is prevalent, people can easily get reinfected by coming in contact with contaminated water. And children, who like to play in water, tend to have the highest burden of the disease.”
For the current paper, Nguyen focused on how global climate change and rising water temperatures might affect each stage of the schistosomiasis transmission cycle. It was already established that both the parasites and the snails are sensitive to water temperature, with each stage having an optimum temperature.
“I wanted to build on previous work to see if we could use it to find better predictors for human risk and more effective interventions,” Nguyen says.
The researchers integrated an epidemiological model of schistosomiasis and temperature-dependent traits of the parasites and their snail hosts to run different computer-simulated interventions. The results showed that interventions targeting snails were most effective at reducing transmission, and pinpointed the water temperature for when the risk of transmission peaks.
Unexpectedly, the simulations also showed that interventions targeting snail removal actually raised the peak transmission temperature by 1.3 degrees centigrade, while reducing transmission risk.
“That may not sound like a lot,” Nguyen says, “but we’re talking about water temperature, which takes a lot of energy to warm, so 1.3 degrees is actually a big shift.”
Snails naturally start to die off at higher water temperatures. The data in the new paper shows how implementing snail control measures, such as through chemical treatment of the water, amplifies snail mortality at all temperatures. This lowers transmission risk overall, but allows peak transmission risk to occur at higher temperatures.
These insights can guide public health workers to time their interventions, by factoring in regional water temperatures, and how the temperatures fluctuate during different seasons of the year.
“Our findings don’t mean that we should stop human treatment for schistosomiasis,” Nguyen says. “Instead, it will likely be beneficial to include both the human and ecological components. By combining human drug treatment with snail removal measures, during times when water is below the peak transmission temperature, we may be able to maximize the efficacy of an intervention.”
Additional authors of the PNAS paper include Jason Rohr (University of Notre Dame), Valerie Harwood (University of South Florida), Rachel Hartman (Emory staff) and Emory graduate student Sandra Mendiola.
The work was funded by the National Institutes of Health, the National Science Foundation, the Porter Foundation and the U.S. Department of Agriculture.