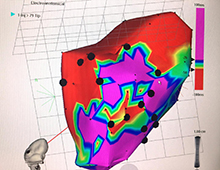
3-dimensional electrochemical mapping was used to target ixmyelocel-T cell therapy to damaged areas of hearts in patients with end-stage heart failure.
Photo by Matthew Brobeck, University of Utah
Patients with heart failure who received an experimental stem cell therapy experienced a reduced rate of death, hospitalization and unplanned clinic visits over the next year compared to a placebo group, according to results presented at the American College of Cardiology meeting in March in Chicago.
The results of the ixCELL-DCM study were published online by The Lancet. It was reportedly the largest cell therapy study done in patients with heart failure so far (58 treated vs 51 placebo).
Emory University School of Medicine investigators led by Arshed Quyyumi, MD, and their patients participated in the study, and Emory was one of the largest enrolling sites. Lead authors were Timothy Henry, MD of Cedars-Sinai Heart Institute in Los Angeles and Amit Patel, MD of the University of Utah.
“For the first time, a clinical trial has shown that administration of a cellular therapeutic results in an improvement in cardiac outcomes based on a pre-specified analysis,” an editorial accompanying an editorial accompanying the paper in The Lancet says.
This study, which was sponsored by Vericel Corporation, was phase II, meaning that a larger phase III study will be needed before FDA approval.
Heart failure is often treated with drugs. However, patients who have class III or IV heart failure symptoms, despite optimal medical treatment, now have few options outside of a heart transplant or a left ventricular assist device (LVAD).
To be part of the study, patients had to have ischemic cardiomyopathy, the most common cause of heart failure, and an implanted cardioverter defibrillator (ICD), and be ineligible for other procedures such as bypass surgery. Most patients had experienced a previous heart attack and were taking several medications such as statins, beta-blockers, ACE inhibitors and diuretics.
In the ixCELL-DCM study, which began in 2012, doctors removed some of each patient’s own bone marrow and sent it to Vericel’s laboratory in Michigan, where it underwent a two-week process to selectively expand two types of cells: mesenchymal stem cells and anti-inflammatory macrophages. Researchers think that these cells may help the heart remodel and function better, but they do not directly rebuild the heart muscle.
The cells were introduced into the patient’s heart with a catheter. The target tissue was selected based on imaging of the border between viable and non-viable heart muscle, according to the rationale and design of the study, published in March in Cell Transplant (open access).
Doctors from 31 sites followed 109 patients over a 12-month period. The 58 patients who received bone marrow cells showed a 37 percent lower rate of deaths, hospitalizations and unplanned clinic visits for symptoms such as fluid buildup and shortness of breath compared to the 51 patients who received a placebo.
Four of the patients who received bone marrow cells died compared to eight in the placebo group. Two patients who received bone marrow cells had an LVAD implanted and died following the implant.
Twenty-two patients (38 percent) who received bone marrow cells were hospitalized with cardiovascular issues during the study while 24 (49 percent) of those who received placebo were hospitalized. There were fewer adverse events, such as those related to catheterization or injection procedures, in the bone marrow treated group compared to the placebo group.
Investigators also monitored structural changes in the heart such as left ventricular ejection fraction (a measure of the heart’s pumping capacity) as well as a patient’s ability to walk for six minutes, but did not see significant differences in either of these measures. Effects beyond one year were not reported. The most recent patient’s 12 month follow-up was in February 2016.
