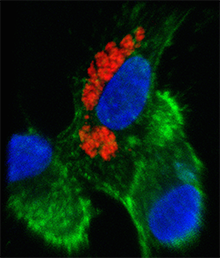
Zika virus can infect and replicate in immune cells from the placenta, without killing them, scientists have discovered. The finding may explain how the virus can pass through the placenta of a pregnant woman, on its way to infect developing brain cells in her fetus.
The results are published in Cell Host & Microbe.
Our results substantiate the limited evidence from pathology case reports," says senior author Mehul Suthar, PhD, assistant professor of pediatrics at Emory University School of Medicine. "It was known that the virus was getting into the placenta. But little was known about where the virus was replicating and in what cell type."
Scientists led by Suthar and Emory pediatric infectious disease specialist Rana Chakroborty, MD, found that Zika virus could infect placental macrophages, called Hofbauer cells, in cell culture. The virus could also infect another type of placental cell, called cytotrophoblasts, but only after a couple days delay and not as readily. Other researchers recently reported that syncytiotrophoblasts, a more differentiated type of placental cell than cytotrophoblasts, are resistant to Zika infection.
The cells for the experiments were derived from full-term placentae, obtained from healthy volunteers who delivered by Cesarean section. The level of viral replication varied markedly from donor to donor, which hints that some women’s placentae may be more susceptible to viral infection than others.
“Not every pregnant woman who is infected by Zika transmits the virus to her fetus,” Suthar says. “Host genetics and non-viral factors, including nutrition and microbiota, as well as timing may be influencing infectivity. A better understanding of these factors could allow the design of preventive measures, and eventually antiviral therapies."
The viral strain used for the experiments was from Puerto Rico, closely related to the currently circulating in Brazil.
For viruses related to Zika (flaviviruses) such as dengue virus, West Nile virus and yellow fever virus, it is rare for the infection to be transmitted from mother to fetus. This is thought to be because of the protective role of the placenta, which separates the circulatory systems of the mother and fetus.
“Zika may be unique in its ability to infect placental cells and cross the placental barrier, in comparison with other flaviviruses,” Suthar says.
It is still possible that disruption of the placental barrier -- as seen in a recent case report -- or antibody-mediated mechanisms are contributing to placental infection by Zika virus, he says.
Hofbauer cells are fetally derived, in contrast with other immune cells found in the placenta called decidual macrophages, which are maternally derived. They are thought to be more tolerant and less prone to inflammatory responses than other immune cells, based on previious work from co-authors Chakraborty and Erica Johnson, PhD.
However, Suthar’s team was able to detect signs of antiviral and inflammatory responses in infected Hofbauer cells. He says his laboratory will continue to investigate Hofbauer cells and their immunology, using both human donated cells and animal models.
“We need to answer questions such as: what are the receptors that allow the virus to enter Hofbauer cells,” he says. “Do these cells change in their immune status during the different phases of pregnancy pregnancy?”
The first authors of the paper are graduate students Kendra Quicke and James Bowen. Additional Emory co-authors include Jens Wrammert, PhD, Bassam Rimawi, MD, Bali Pulendran, PhD, and Raymond Schinazi, PhD.
Research in Suthar’s laboratory is supported by the National Institute of Allergy and Infectious Diseases (U19AI083019, R56AI110516, R21AI113485).
