How does a protein evolve a new function while keeping its old function? How do RNA molecules emerge from obscurity to acquire important roles in cell regulation?
The W.M. Keck Foundation has awarded Emory a five-year $1 million grant to support the work of biochemist Eric Ortlund, PhD and his lab in examining these questions as they relate to the glucocorticoid receptor, part of the family of steroid hormone receptors.
The Keck Foundation, established in 1954 by Superior Oil founder William Myron Keck, supports community service projects in Southern California as well as science, engineering and medical research and undergraduate education across the country.
The Emory project could inform anti-inflammatory and anti-cancer drug discovery efforts as well as deepening scientists’ understanding of how evolution takes place on a molecular level.
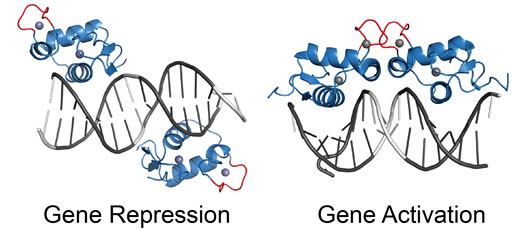
Ortlund’s project focuses on the glucocorticoid receptor, the protein target for the stress hormone cortisol as well as synthetic anti-inflammatory drugs such as prednisone and dexamethasone. The glucocorticoid receptor turns some genes on and others off, and binds DNA in two different modes to accomplish these distinct functions. The glucocorticoid receptor’s relatives, such as the progesterone and mineralocorticoid receptors, only have the activating function.
Part of the Ortlund lab’s project is to decipher how the glucocorticoid receptor’s second function emerged, step by step, by analyzing the structure of “resurrected” steroid receptor molecules. These are ancestral proteins that do not exist in any living organism, but whose sequence and structure can be reconstructed with high confidence.
The glucocorticoid receptor is also the target of a decoy RNA molecule called Gas5 that restrains its activity and is a key player in some forms of cancer. Ortlund’s lab will also examine how the interaction between Gas5 and the glucocorticoid receptor evolved.
“We will walk these ancient proteins and non-coding RNAs forward through evolutionary time to understand how DNA sequence changes drove changes in cell signaling,” Ortlund says.
The project is expected to include collaborators Graeme Conn, PhD at Emory as well as Eric Gaucher, PhD at Georgia Tech, Douglas Kojetin, PhD at Scripps Research Institute and Ivaylo Ivanov, PhD at Georgia State.