Learning from Lampreys
An ancient immune system provides insights into our own

Lampreys are described as primitive predators, aggressive flesh feeders, and piston-tongued parasites. To watch a lamprey attach itself to the glass of an aquarium or dangle off the side of an unfortunate fish causes an involuntary shudder.
In the Great Lakes region, lampreys are viewed as fish-killing pests and an invasive species and are regularly poisoned to control their numbers. In Europe during the Middle Ages they were considered a delicacy.
But in immunologist Max Cooper’s Emory Vaccine Center lab, researchers see lampreys as windows into the distant past and biomedical treasure troves that may contain disease-fighting secrets from another eon.
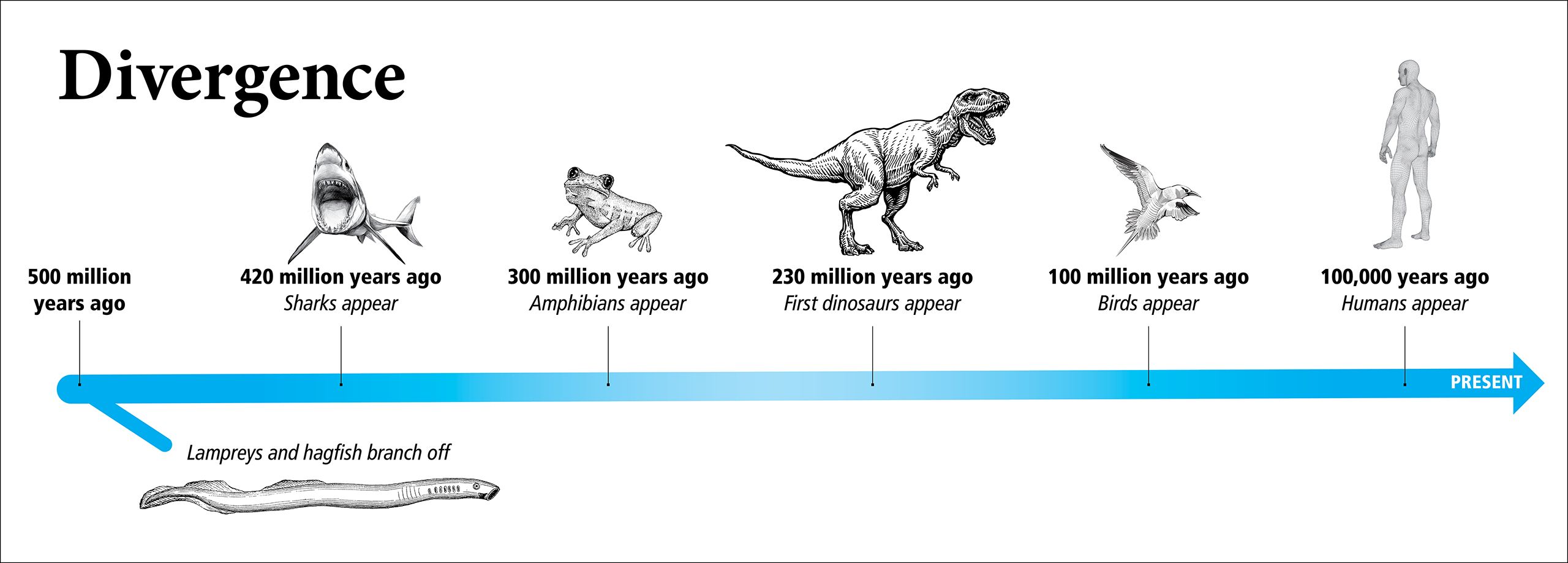
Lampreys and their close cousins, hagfish—both jawless vertebrates—are early offshoots on the evolutionary tree, having diverged from jawed fish and sharks and, ultimately, humans, hundreds of millions of years ago.
“Our studies led to the discovery that two very different types of adaptive immune systems have evolved in vertebrates,” says Cooper. Adaptive immune systems can “remember” and target certain pathogens they have encountered before.
While lampreys may look primitive, like parasitic tubes with teeth, they have sophisticated immune systems, which is why Cooper is interested in them.
To defend themselves against microbes, lampreys’ immune cells produce proteins that grab onto foreign substances, much the same as our own antibodies. But these proteins don’t look anything like the antibodies found in mammals, birds, and jawed fish.
So studying these creatures provides scientists a glimpse of where the human immune system came from—as well as how ours might have looked if evolution had taken a different path.
While vertebrates went on to develop jaws, and eventually evolved into mammals, primates, and humans, vertebrates without jaws (lampreys and hagfish) stayed similar in form to their genetic forebears.
Mature lampreys attach themselves to fish and feed on their innards or their blood. While adults can extend as long as your arm, Cooper’s lab studies the finger-sized larvae. In the wild, these lamprey larvae feed on the muck at the bottom of streams and rivers for a few years before getting big enough to go foraging. “We work with the juveniles, which are like teenagers if humans became adults at age 50,” says Jonathan Rast, senior scientist.
In Emory’s animal facilities, scientists keep lampreys in tubs and feed the larvae brewer’s yeast. Cooper’s lab gets its lampreys from biologists in the Midwest, who catch lampreys by first stunning them with electricity.
“We used to do the same kind of thing when I was growing up in Mississippi,” Cooper says. “It was called ‘telephone fishing’ because someone would crank the magnet from a telephone.”
The story of why lampreys came to attract Cooper’s attention, however, begins more than five decades ago—not with fish, but with fowl.
Invader Alert
Two main types of white blood cells, or lymphocytes, drive our adaptive immune responses: T cells, which use cell-to-cell contact, and B cells, which produce antibodies.
T cells shuttle through our bodies ready to respond to invaders—think K-9 sniffers. Some T cells can target and kill cells that are infected by viruses because tiny traces of the invader are found on the target cells’ surfaces.
In contrast, B cells are like factories designed to make bombs (antibodies) that will blow up one specific enemy.
We know about B cells and T cells because of pioneering work done by Cooper in the 1960s with Robert Good at the University of Minnesota that helped lay the foundation for modern immunology.
Cooper started his career as a pediatrician and became interested in children with inherited immunodeficiencies. One group of kids had trouble fighting viruses, while others had trouble making antibodies.
This led him to study birds, because in the 1950s, researchers at Ohio State had already had some success modeling antibody deficiency in chickens.
B is for bursa
B cells are named after an organ found only in birds, called the bursa of Fabricius. T cells are named after the thymus, where they develop in both birds and mammals.
Cooper and Good were the first to demonstrate that the B cells needed for antibody production develop in the bursa, and that B cells and T cells develop in different places.
“The effect of this accomplishment is difficult to overstate,” wrote Alexander Gitlin and Michel Nussenzweig in the journal Nature. “Through their discovery of the B and T cell systems, Cooper and his colleagues set in motion a series of landmark findings… As these discoveries continue to affect human health, it remains important to remember their origins in an experiment performed 50 years ago, by Cooper, in chickens.”
In humans, the precursors of B cells and T cells begin in the bone marrow but develop along different paths, reuniting in the spleen and lymph nodes, where they collaborate to mount immune responses and take down pathogens.
In the 1970s, the discovery of B cells gave birth to monoclonal antibody technology. Basically, this allowed researchers to make clones of antibodies that target particular cells or proteins.
Over the past few decades the FDA has approved more than a dozen monoclonal antibodies to treat cancers. Because they can more specifically target cancer cells, antibodies aren’t as destructive to healthy cells as chemotherapy or radiation.
More recently, monoclonal antibodies have been enlisted to fine-tune the immune system. The drug that enabled former President Jimmy Carter to beat metastatic melanoma? A monoclonal antibody.
Researchers have created antibodies for even more diseases, such as inherited high cholesterol, rheumatoid arthritis, multiple sclerosis, and Ebola.
Disease-fighting clues
Cooper had begun studying lampreys and their immune systems to figure out which came first, B cells or T cells. The secondary benefits of this research are still unfolding: What kinds of disease-fighting clues and cures might lamprey antibodies contain?
Lampreys’ antibody-like molecules are called “variable lymphocyte receptors” or VLRs.
When Cooper and colleagues first discovered VLRs at the University of Alabama-Birmingham, the scientists recognized that they might also have potential as diagnostic tools or cancer-fighting drugs.
When Cooper came to Emory in 2008, his team continued to explore how to harness lamprey antibodies’ unique properties. VLRs could offer some advantages over standard, mammalian antibodies that are currently used against cancer and autoimmune diseases. As proteins, VLRs are sturdy and don’t need to be refrigerated. And they are simple—they come in one piece and are easier to engineer genetically.
“In every case we’ve tested, VLRs were more specific than a standard antibody,” Cooper says, citing experiments in which lamprey antibodies were isolated after the lampreys were immunized with anthrax spores. “Even when VLRs recognize the same target as a conventional antibody, they recognize it differently. I think it’s because their structure is simpler and more rigid.”
And possibly, a whole lot older.
500 million year advantage
Lamprey antibodies are relative strangers to certain modern pathogens. When fighting disease, this lack of antibody familiarity allows doctors a leg up.
Because VLRs come from vertebrates so far removed from humans, they can be used to target important molecules that have persisted in similar form through millions of years of evolution—so-called “conserved antigens.”
Our immune system generates vast libraries of potential antibodies, but the ones that react against “self”(something already in the body) are generally muted or edited out. If they weren’t, our immune system would be in perpetual overdrive, similar to what occurs in autoimmune diseases.
If researchers are trying to make antibodies against something in mice, they can get around this muting response by immunizing and collecting antibodies from different animals—say, rabbits or goats—instead. But what if they are dealing with a molecule that’s common to almost all mammals? That’s where lampreys come in. “It’s hard to generate a classical monoclonal antibody against a conserved antigen,” Cooper says. “But if you are separated by 500 million years of evolution, like lampreys are, you may have a better chance.”
One example of an immune target that has been difficult is plasma cells, the bone marrow cells that churn out antibodies. In a sense, plasma cells are specialized, fully mature B cells; the healthy counterparts to multiple myeloma cells.
Former Cooper lab member Goetz Ehrhardt, now at the University of Toronto, immunized lampreys with bone marrow from a multiple myeloma patient. Scientists were able to pick out a VLR that reacted specifically with plasma cells and myeloma cells. The VLR they found targets the same molecule as a recent FDA-approved treatment for multiple myeloma—proof that lamprey antibodies could be useful.
“Because of the unique origins of lamprey antibodies and their radically distinct protein architecture, these molecules may be able to recognize structures that conventional antibodies cannot,” Ehrhardt says.
Weaponizing antibodies
A biotech company called NovAb, formed with the help of the Georgia Research Alliance, aims to commercialize lamprey antibodies. NovAb created a “weaponized” version of the same antibody that recognizes plasma cells and has shown good results in the lab. In mice, this version works just as well as standard treatments, says company CEO Ed Cannon.
In Emory’s Department of Pediatrics, Associate Professor Trent Spencer and graduate student Robert Moot have also been experimenting with lamprey antibodies for possible use against leukemia or neuroblastoma, a common form of tumor in children.
Looking ahead, Cannon and Cooper say that lamprey-derived molecules may have special roles to play that ordinary antibodies can’t. “We are not interested in reproducing or replacing existing drugs,” Cannon says. “We are more interested in targeting molecules against which conventional antibody technology has been often unsuccessful.”
One possible target: brain cancer stem cells.
While the potential anticancer weapons proliferate, Cooper’s lab continues to unearth more of lampreys’ basic biology.
Despite the progress of the last decade, how lampreys slice and dice their genes to create a huge variety of antibodies is still mysterious.
And, perhaps because of lampreys’ status as pests, little is known about the viruses that lampreys’ immune systems are called upon to fight. “Viruses played a huge role in shaping the evolution of the vertebrate immune system,” says virologist and cell death expert Ed Mocarski, an Emory Woodruff Professor in microbiology and immunology. “If more is known about viruses that infect lampreys, it could unlock other molecular details about lamprey immunology.”
Lamprey antibody technology also could be applied against HIV, influenza, and other infectious agents.
Although ancient in origin, lampreys could become the foundation for the next generation of immunotherapy weapons, able to boost our body’s own ability to fight disease.
That’s enough to make these toothy parasites seem downright valiant.
“One of the most remarkable things about a career in biomedical research is that you can start almost anywhere and end up in the most unforeseen places, being constantly amazed by what you are learning along the way.”—Max Cooper
On Becoming a Physician Scientist
Little did I imagine while growing up in rural Mississippi in the 1930s that I would eventually pursue a research career in immunology. My childhood was rich in unfettered time, space to roam freely, woods and streams to explore, an uncontaminated view of starry night skies to ponder, and an abundance of books to read. The treasure of books and the thirst to read them were gifts of my father, a mathematician and educator, and my mother, also a teacher. We lived on the campus of the 12-grade school where my father was the superintendent.
The tranquility of life was abruptly altered in 1941 by U.S. entry into World War II. Older boys in the community were drafted into the military services, my mother began to work in an underground munitions plant, and, in 1945, my older brother joined the Marines at age 17, just before peace was declared.
As a high school senior obsessed with sports, hunting, and girls, I began to realize that reaching my stated goal of becoming a doctor would require a great zeal for study and a long time in school at a high cost.
The tragic death of my wonderful and adventurous older brother in an automobile accident abruptly changed this equation for me. He had made me the beneficiary of his service insurance policy.
During this time of intense family grieving, my father took me aside to tell me that I now must do what my brother and I both would have done. Although my interests did not change overnight and my study habits failed to undergo any miraculous transformation, I began the well-prescribed course to becoming a doctor.
My fascination with caring for patients and understanding the pathophysiology of their diseases grew at each step along this career path. It became more and more obvious that I had stumbled into an endless wonderland of challenging problems.
This is an excerpt from an essay by Max Cooper that first ran in the Annual Review of Immunology.
Windows of Opportunity
Donors who support early stages of pioneering research receive a significant return on investment. For every $1 donated, the Emory Vaccine Center secures $17 in federal grants.
To find out more, contact Ashley Michaud, senior director of development, at 404.778.1250 or ashleymichaud@emory.edu.